Duke Cancer Institute investigators Dorothy Sipkins, MD, PhD, associate professor of medicine, pharmacology and cancer biology, and P. Kelly Marcom, MD, professor of medicine, have just launched a proof-of-concept clinical trial of a new drug for hormone receptor positive breast cancer patients whose disease has spread to the bone.
The investigational therapy, GMI-1359, targets both E-selectin and CXCR4, normal inflammatory molecules that also appear to play a role in tumor trafficking and metastasis to the bone.
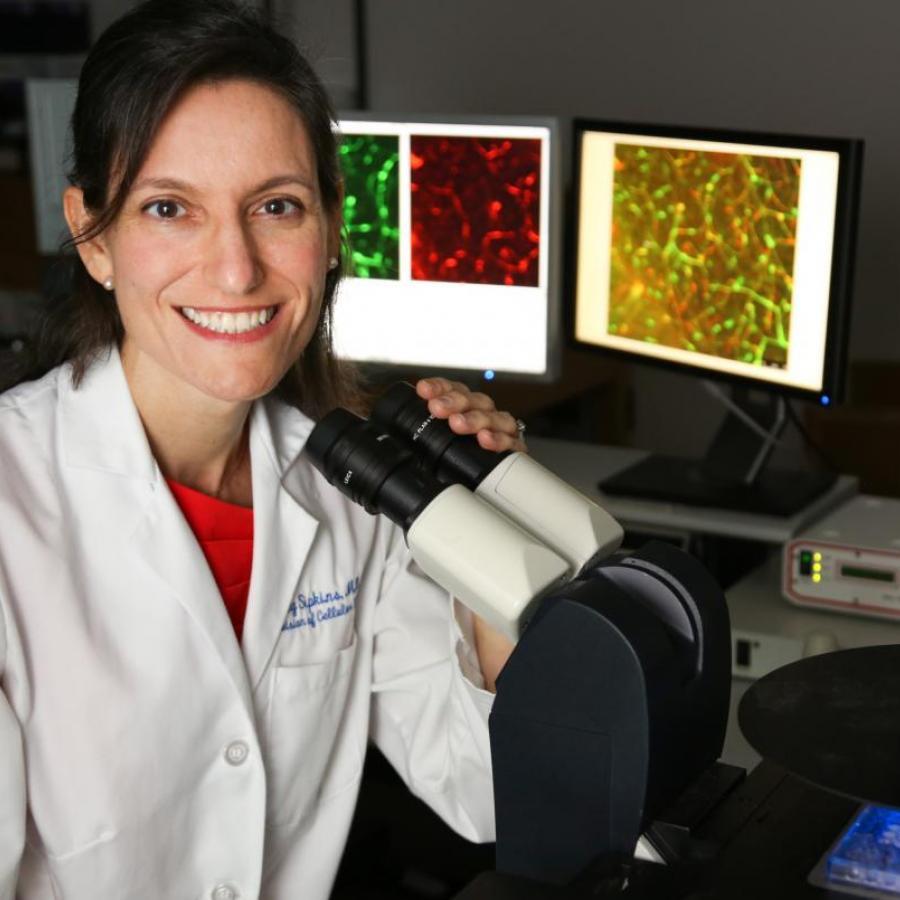
The drug, discovered and invented by glycobiologist John Magnani, PhD, Senior Vice President and Chief Scientific Officer at GlycoMimetics, Inc., intersects with years of preclinical research by Sipkins, first in leukemia and then in breast cancer.
The new therapy, which co-principal investigators Sipkins and Marcom are calling “a micro-environment targeted therapy,” has the potential to improve treatment of patients at risk of cancer metastasis to the bone, or whose cancer has already “micro-metastasized” to protective pro-dormancy niches in the sinusoid capillaries (sinusoids) of the marrow.
“The Sipkins therapy, for short,” said Marcom, crediting Sipkins’ “world class bench science and translational work.”
“John Magnani has been researching E-selectin for years,” said Sipkins. “My work showing the role of both E-selectin and the SDF-1 receptor, CXCR4, in leukemia cell trafficking to the bone marrow [published in Nature, 2005] intrigued him, and he considered the possibility that a drug that inhibited both molecules might be very useful.”
In that study, Sipkins showed that CXCR4 (with its chemokine SDF1) and E-selectin played a role in trafficking and adhesion of leukemic cells into the sinusoids.
After her discoveries in leukemia, she pivoted to breast cancer.
“I became obsessed with these two molecules, E-selectin and CXCR4,” said Sipkins. “The first question we had was if breast cancer cells were using the same gateways to get into the bone marrow.”
When she learned that breast cancer cells, unlike leukemic cells, didn’t need CXCR4/SDF1 to enter the sinusoids, she become more focused in on E-selectin.
Sipkins then asked Magnani for access to a drug he was developing in the hematology space to inhibit E-selectin — GMI-1271/uproleselan.